Fetal Stem Cells and Autism: A Treatment the World Rarely Talks About
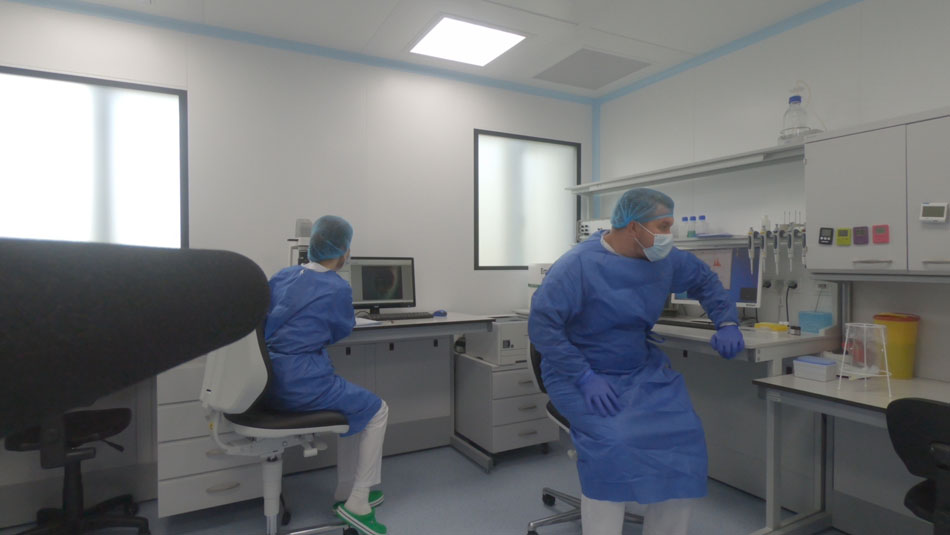
In September of 2019 I met two American families in Kyiv, Ukraine. Both had traveled across the world seeking treatment for their sons, who had been diagnosed with autism.
Seven-year-old Matt had struggled for years with communication and emotional regulation. Four-year-old Jax had experienced speech regression around eighteen months and had become largely non-verbal. Like many parents facing an autism diagnosis, both families had already tried speech therapy, occupational therapy, and specialized educational programs.
Yet they had traveled halfway across the world to pursue something almost no one in the United States has ever heard of: fetal stem cell therapy.
At the time, neither family knew the other. By coincidence they arrived at the same clinic in Kyiv, both searching for the same possibility — improvement in their children’s ability to communicate, interact, and live more independent lives.
Several months after their treatment, I began receiving text messages from both families describing changes they were witnessing in their children.
The improvements were significant enough that both families later returned to Kyiv in 2021 for a second round of therapy — this time meeting each other and going through the process together.
The 17-minute video below tells their story. Scroll to the end of the article for a March 2026 update on both families.
Why Fetal Stem Cells Are Different From Other Stem Cell Therapies
Most people who have heard of stem cell therapy are actually familiar with a very different category of cells known as mesenchymal stem cells (MSCs).
MSCs are commonly derived from sources such as:
- Bone marrow
- Umbilical cord tissue
- Placenta
- Adipose (fat) tissue
While these cells are useful for certain regenerative purposes, they represent only one category of stem cell.
Mesenchymal stem cells do not contain the wide range of specialized progenitor cells found in fetal stem cell preparations, particularly those associated with the neurological system. For this reason, MSC therapies are generally used for inflammation control and tissue repair, rather than targeting neurological development.
This distinction is important because MSC therapies are currently the most widely available stem cell treatments in the world. They are derived from sources such as bone marrow, placenta, umbilical cord tissue, or adipose tissue.
As a result, many clinics advertise MSC treatments for a wide range of conditions — including autism — largely because MSCs are the only category of stem cell widely permitted for clinical use in most countries.
Fetal stem cell therapy, by contrast, delivers a much broader spectrum of developmental progenitor cells, including those related to neurological tissues. Today, the only clinic legally producing and administering these fetal stem cell preparations is EMCELL in Kyiv, Ukraine.
Rather than delivering a single category of cell, fetal stem cell preparations contain a wide spectrum of specialized progenitor cells that correspond to many of the body’s major tissues and organ systems.
This includes cells involved in the development of:
Blood and immune systems
- Hematopoietic stem cells (precursors to bone marrow and blood)
Major organs
- Hepatic stem/progenitor cells (liver)
- Renal stem/progenitor cells (kidney)
- Adrenal stem/progenitor cells (adrenal glands)
- Pancreatic beta progenitor cells (pancreas)
Cardiovascular system
- Endothelial stem cells (blood vessels and capillaries)
- Cardiac stem/progenitor cells (heart)
Respiratory and digestive systems
- Lung stem/progenitor cells
- Intestinal stem/progenitor cells
Musculoskeletal tissues
- Myosatellite stem cells (muscle)
- Chondrogenic stem/progenitor cells (cartilage)
- Connective tissue progenitor cells
Neurological system
- Neuronal stem/progenitor cells
- Immature post-mitotic neurons
- Dopamine progenitor cells
- Oligodendrocytes (central nervous system support cells)
- Glial cells (critical for brain signaling)
In addition, fetal stem cell preparations contain growth factors and neuromediators that promote cellular signaling and tissue regeneration.
One of the most important distinctions is that many of these cells can interact with the central nervous system, including cells capable of influencing neurological repair and development. This characteristic is one of the reasons researchers began exploring fetal stem cells for neurological conditions such as autism, cerebral palsy, and neurodegenerative disorders.
One of the most significant biological characteristics of fetal stem cells is their natural role in building the developing nervous system. During fetal development, progenitor cells migrate throughout the body and into the brain as organs and neural networks form. These cells possess unique signaling and migratory properties that allow them to cross biological barriers, including the blood-brain barrier. Because of this developmental behavior, fetal-derived cells have long attracted scientific interest for conditions involving the brain and central nervous system.
Below is a graphic illustrating all of the cells provided to most every patient at EmCell.

Video: Why Fetal Stem Cells Are Different From Other Stem Cell Therapies
Why the Comparison to Other Stem Cell Treatments Matters
Because MSC therapies dominate the global stem cell industry today, many people assume all stem cell treatments are essentially the same.
They are not.
Mesenchymal stem cell therapies involve one category of multipotent cell, typically derived from bone marrow, umbilical cord tissue, placenta, or adipose tissue. These cells are most commonly used for immune modulation and tissue repair signaling.
Fetal stem cell therapy is fundamentally different.
Rather than delivering a single type of stem cell, fetal stem cell preparations contain a broad spectrum of specialized progenitor cells corresponding to multiple organ systems of the body, including cells associated with neurological tissues.
This difference in biological complexity is significant.
While MSC therapies provide one category of regenerative cell, fetal stem cell preparations deliver a much wider developmental toolkit of cells involved in the formation of blood, immune, organ, and neurological systems.
For this reason, comparing MSC therapy to fetal stem cell therapy is not truly an apples-to-apples comparison.
As some physicians familiar with the field have described it, the difference can feel less like comparing two similar technologies and more like comparing a horse and carriage to a jet airplane.
This fundamental difference is one of the reasons fetal stem cell therapies have attracted both significant scientific interest and intense regulatory controversy.
A Medical History That Began With Chernobyl
The modern clinical use of fetal stem cells traces back to one of the most devastating disasters in human history: the 1986 Chernobyl nuclear meltdown.
In the aftermath of the explosion, many victims exposed to high levels of radiation developed aplastic anemia, a condition in which the bone marrow is severely damaged and can no longer produce blood cells. At the time, physicians faced a dire medical crisis. Patients whose bone marrow had been destroyed by radiation often could not survive without a compatible bone marrow transplant — something that was rarely available quickly enough.
A group of Soviet scientists and physicians in Ukraine, who would later become the founders of what is now EMCELL, began experimenting with an alternative approach using fetal liver cells. These cells are rich in hematopoietic stem cells responsible for forming the body’s blood and immune systems during early human development.
In several cases, these fetal cells were able to restore blood production without requiring a traditional bone marrow transplant. These early treatments represented one of the first documented clinical uses of fetal-derived stem cells in modern medicine.
The work attracted international medical attention, including coverage in the Los Angeles Times in May 1986, which reported on the use of fetal liver cells to help treat victims suffering from radiation-induced bone marrow failure.
Following these early successes, the same group of Ukrainian physicians and researchers continued developing fetal stem cell therapies in the decades that followed. Their work eventually expanded beyond radiation injuries to include applications for immune disorders, degenerative diseases, and neurological conditions.
Today, that original line of research continues at EMCELL in Kyiv, Ukraine — the only laboratory and clinic currently licensed to produce and administer fetal stem cell therapies.
Video: A Brief History of Fetal Stem Cells
Why Autism Became a Focus
Autism spectrum disorder (ASD) is a complex neurological condition that affects communication, behavior, and social interaction. Its exact causes remain debated and likely involve multiple biological factors including immune dysfunction and neurological development.
Because stem cells have the ability to influence immune regulation and cellular repair, some physicians began exploring whether regenerative medicine might help improve neurological function in autistic children.
This is where fetal stem cells entered the conversation.
Unlike many experimental stem cell approaches, fetal stem cells are already highly specialized developmental cells, capable of signaling tissue repair and immune modulation. These characteristics are why some researchers believe they may have therapeutic potential for neurological conditions.
How the Autism Treatment Protocol Works
Children receiving fetal stem cell therapy for autism at EMCELL are treated using a protocol designed to target both immune regulation and neurological function.
The therapy typically involves multiple delivery methods, each intended to reach different biological systems.
According to the treatment protocol described in the video:
- Intravenous infusion
Patients receive fetal stem cells intravenously containing a wide array of immunological and hematopoietic stem cells. These cells circulate throughout the body and are believed to support immune system regulation and systemic repair processes. - Subcutaneous injection
Additional stem cells — including neuronal and neurological progenitor cells — are administered subcutaneously. This allows gradual absorption and interaction with surrounding tissues. - Intranasal delivery
Neurological stem cells are also delivered intranasally, allowing cells to travel along olfactory pathways toward the brain and bypass portions of the blood-brain barrier.
This delivery method is one reason fetal stem cell therapy has been explored for neurological conditions, including autism.
EMCELL’s Reported Clinical Experience with Autism
According to clinical data released by EMCELL, the clinic has performed:
1,155 treatments (including some children with related conditions such as ADHD)
715 children diagnosed with autism
Patients from multiple countries, including the United States
Of the 781 parental feedback reports collected, approximately:
76.1% reported positive changes in behavior or overall condition.
More recently, from 2019–2025, the clinic reports:
86.4% positive feedback following treatment protocol improvements.
It is important to note that the statistics referenced in the video were compiled several years ago. EMCELL continues to update its clinical data regularly. For readers interested in the most current outcome statistics and detailed data analysis, the latest published statistics document is provided here.
The clinic states that improvements commonly reported by parents include:
Better communication and speech development
Improved social interaction
Increased emotional responsiveness
Better sleep patterns and behavior regulation
Like many therapies for autism, evaluation often relies on behavioral scales and parental observations, including tools such as the Autism Treatment Evaluation Checklist (ATEC).
Click here to view, print, or download the entire report in PDF form.
EMCELL’s Published Data on Autism
— Clinical Case of Effective Fetal Stem Cells Treatment in a Patient with Autism Spectrum Disorder (2025)
— Clinical Case of Successful Therapy for the Patient with Autism by use of Fetal Stem Cells (2024)
— Efficacy of Fetal Stem Cell Transplantation in Autism Spectrum Disorders: An Open-Labeled Pilot Study (2014)
— Perspectives on the Use of Stem Cells for Autism Treatment (2013)
— Efficacy of Fetal Stem Cell Transplantation in Autism Spectrum Disorders: A Pilot Study (2013)
Why This Therapy Exists in Only One Place
One of the most striking realities surrounding fetal stem cell therapy is that it is illegal in most countries — with Ukraine standing as the only place where it is legally produced and administered in a clinical setting.
In the United States and much of Europe, strict regulatory frameworks surrounding fetal tissue research have effectively prevented this therapy from developing into a widely studied clinical treatment.
Ukraine, however, established a legal framework decades ago allowing controlled medical use of fetal stem cells, which enabled EMCELL to continue its research and clinical work.
As a result, families from around the world — including the United States — have traveled to Kyiv seeking treatment that simply does not exist anywhere else.
Video: USA/EU Regulation issues; Fetal Stem Cell Production & Quality Control at EMCELL
A Personal Note
As a filmmaker and investigator, my role has never been to promise cures or advocate blindly for controversial medical treatments. My goal has always been to document what exists, ask difficult questions, and allow people to see the evidence for themselves.
When I began documenting fetal stem cell therapy in 2014, I met families who were willing to travel across the world in search of something that conventional medicine could not provide them.
Over time, many patients began asking me a simple question: “You’re making a documentary about this… and you haven’t tried it yourself?”
Given that this therapy is also explored in areas of preventative and regenerative medicine — not only for those with serious illness — I ultimately chose to experience it firsthand. Since 2014, I have personally undergone fetal stem cell therapy nine times.
What is undeniable is that this therapy — its history, its science, and the patients pursuing it — deserves to be discussed openly and honestly.
The video below includes footage of my own treatment. Or, watch the entire feature documentary here.
March, 2026 Update on Matt and Jackson
From the Lippincott family: (March, 2026)
Our trip to EmCell in 2019 changed our family’s life.
At the time, Jackson was a 4-year-old nonverbal autistic boy. He relied on a tablet to communicate, walked on his toes, struggled with severe sensory issues, and had an extremely limited appetite. The challenges felt endless.
Today, Jackson is 10 years old—and our lives look completely different.
He speaks in full sentences. He no longer toe walks. He’ll try any food at least once. His sensory issues have faded. He has friends, plays organized sports, and is thriving academically in school.
Every day, we are reminded of how far he’s come.
We are forever grateful for our miracle boy.




From the Cutter family (March, 2026):
Matt repeated 6th grade and is now in 7th grade with a full-time one-on-one aide.
He absolutely loves sports and recently played on a basketball team—something that would have been unimaginable just a few years ago, when the stimulation would have been overwhelming for him. The rules can still be challenging at times, but the fact that he’s out there playing is something we don’t take for granted.
He also loves to run, swim, and—most of all—he truly enjoys going to school, which means so much to us.
We think of you both often. We miss you and hope you’re doing well.

About the Author
Eric Merola is an internationally award-winning documentary filmmaker known for exploring controversial and often overlooked areas of science and medicine. Over the past decade, he has written, directed, and produced multiple feature-length documentaries examining disruptive medical technologies and the institutions that challenge them.
His work includes the Burzynski film series, Second Opinion: Laetrile at Sloan-Kettering, The God Cells: A Fetal Stem Cell Journey, The Andorra Hustle, and Ukraine: Fetal Stem Cell Pioneers. These films have received international distribution and multiple awards, while also generating significant public discussion around medical innovation and regulatory systems.
Before transitioning into documentary filmmaking, Merola worked as a motion graphics designer and animation director, producing content for networks and media outlets including Comedy Central, A&E, and Nickelodeon.
In addition to filmmaking, Merola is also a painter and visual artist, creating large-scale murals and original works. His artwork can be viewed at ericmerola.gallery.
Through his work, Merola focuses on documenting real-world patient experiences, scientific controversy, and emerging therapies that exist outside of mainstream awareness — with the goal of encouraging open discussion and critical thinking around complex medical topics.






























































Recent Comments